Instructions for authors
INSTRUCTIONS FOR AUTHORS
SALUS – Journal of Health Sciences / Revista de Ciências da Saúde
EMESCAM – Escola Superior de Ciências da Santa Casa de Misericórdia de Vitória
CNPJ 28.141.190/0004-29
Av. Nossa Senhora da Penha, 2190, Santa Luiza, Vitória, ES, CEP 29045 402
E-mail: contato@salusjournal.org
The general publication rules are summarized in the “Uniform Requirements” for Manuscripts Submitted to Biomedical Journals: “Writing and Editing for Biomedical Publication” (www.icmje.org/recommendations). Special suggestions are given to systematic bibliographic reviews and meta-analyzes, retrospective and observational studies on epidemiology, clinical trials, diagnostic accuracy and prospective studies, case reports and case studies. We ask the authors to check the information below before submitting the manuscript to SALUS.
INFORMATIONS FOR AUTHORS
The Salus – Journal of Health Sciences is the scientific magazine by EMESCAM – Escola Superior de Ciências da Santa Casa de Misericórdia de Vitória (School of Sciences of Santa Casa de Misericórdia in Vitória) and it is published every four months.
SALUS aims to publish relevant researches in health science and health policy areas that help social development in a globalized manner. It promotes the study, improvement and update of expert professionals, through the discussion, distribution and the promotion of evidence-based information.
The works submitted to publication in SALUS must concern themes related to health sciences and health policies.
The following article categories are published in the magazine: original article, editorial, review/update article, case report, brief communication and preliminary note, letter to the editor, experimental work, clinical-surgical correlation and multimedia.
The acceptance will be based in originality, significance and scientific contribution. Articles with mere propagandistic or commercial aims will not be accepted.
The authors are responsible for the content and information in their manuscripts.
The magazine will be fully published in its website (www.salusjournal.org).
EDITORIAL POLICY
Style
SALUS adopts the Vancouver Style – Uniform Requirements for Manuscripts Submitted to Biomedical Journals, by the International Committee of Medical Journal Editors, available at: www.icmje.org/recommendations
Submission and Publication Policy
Only manuscripts from which data are not under evaluation by other journals and/or have not been previously published will be considered for review.
The approved manuscripts may only be fully or partly reproduced after express consent by the SALUS’ editor.
Electronic Submission
The manuscripts must be mandatorily electronically submitted at the website (www.salusjournal.org). Once in this link, the system will ask the author’s username and password if he/she is already registered. Otherwise, click the button “I want to sign up” and get registered. Yet, if the author has forgotten his/her password, he/she must use the mechanism to remember your password, which will send an e-mail with the password to the author.
The submission system is self-explanatory and consists of 8 steps:
- 1st Step: article classification
- 2nd Step: inclusion of title and keywords
- 3rd Step: authors’ registration
- 4th Step: Abstract inclusion (in Portuguese and English)
- 5th Step: inclusion of the manuscript with references
- 6th Step: sending pictures
- 7th Step: generating the copyright transfer statement, conflict interest and a copy of the Legal Opinion by the Institution’s Ethic Committee of Research.
- 8th Step: article sending/Submission finishing
The texts must be written in Word format and the pictures and tables must be recorded in separate files.
Registrations must be updated, because the communication with the authors is exclusively done by e-mail.
The authors may follow the status of their works at any time in the website by the publication management system, through the flow code automatically generated by the system, or even by the title of the works. If the article is “non-standard”, the author must be warned by e-mail and may correct it.
Peer review
All scientific contributions are reviewed by the Editor, Associate Editors, Members of the Editorial Council and/or Invited Reviewers. The reviewers answer a small questionnaire, in which they sort the manuscript and make a rigorous examination of all items that must compose the scientific work and assign a score to each item of the questionnaire. In the end, general comments about the work are done and it suggests if the work should be published, corrected – according to the recommendations – or definitely rejected. The editor will make the decision based in these data. In case of discrepancies among the evaluators, a new opinion may be requested in order to get a better judgment. When modifications are suggested, they will be forwarded to the main author and then to the reviewers, so they may check if the demands were attended. The authors have 10 days to perform the modifications demanded by the reviewers and resubmit their articles. The authors must highlight the modifications performed in their texts answering to comments/suggestions by reviewers. Losing this deadline will imply the removal of such articles from the review process.
Once articles are accepted, proofs of edited articles (PDF format) will be sent to their authors for evaluation and final approval.
Language
The articles must be written in Portuguese or English, using scientific, clear and precise language, avoiding the informality of colloquial language. The magazine will provide translation for works that do not have an English or Spanish version.
SALUS publishes articles in Portuguese and English and abstracts in Portuguese, English and Spanish.
Research with Human Beings and Animals
Investigations in human beings must be submitted to the Ethics Committee of the Institution, fulfilling the Declaration of Helsinki from 1975, reviewed in 2008. (World Medical Association, available at: www.wma.net/en/30publications/10policies/b3/17c.pdf), and the Resolution CNS 466/12 – Brazilian National Health Council (Conselho Nacional de Saúde), available at http://conselho.saude.gov.br/resolucoes/2012/Reso466.pdf
Experimental works involving animals must be submitted to the Ethics Committee of Research with Animals, respecting Federal Law n. 11.794/2008, Decree n. 6.899/2009 and CONCEA (National Council for Control of Experimentation with Animals) Resolution n. 12/2013 – Brazilian Guideline of Practice for Care and Use of Animals for Scientific and Teaching Purposes (Diretriz Brasileira de Prática para o Cuidado e Utilização de Animais para fins Científicos e Didáticos – DBCA), available at: http://concea.mct.gov.br.
The randomized studies must follow the guidelines of CONSORT (available at: www.consort-statement.org/consort-statement).
SALUS supports the policies for registration of clinical trials by the World Health Organization (WHO) and by the International Committee of Medical Journals Editors (ICMJE), acknowledging the importance of such initiatives for the international recording and broadcast of information about clinical studies, in open access. Therefore, only clinical research articles that received identification number at one of the Clinical Trial Registrations validated by the criteria established by WHO and ICMJE will be accepted. The addresses are available at ICMJE website (www.icmje.org). The identification number must be registered at the end of the abstract.
The approval statement of the study by the Ethics Committee of Research must be forwarded along with the manuscript submission. Articles that do not demand such approval must be justified.
Copyright Transfer and Conflict of Interest Statements
The authors of the manuscripts must forward the copyright transfer statement along with the submission, signed by all the authors.
All the published manuscripts become permanent property of SALUS and may not be published without the written consent of its editor.
Similarly, a conflict of interest statement must be forwarded to validate the manuscript submission, signed by all the authors.
Both documents, the copyright transfer statement and the conflict of interest statement, are standardized and provided by the system at the moment of manuscript submission.
Authorship Criteria & Individual Contribution for the Research
We suggest that the authorship criteria of the adopted articles follow the recommendations of the International Committee of Medical Journal Editors. Thus, only those who contributed directly to the intellectual content of the work must be listed as authors.
The authors must match all the following criteria, in order to have public responsibility for the content of their work:
- To have created and planned the activities that led to the work, or interpreted the results found, or both;
- To have written the work or revised the successive versions and taken part in the revision process;
- To have approved the final version.
People who do not match the above requirements and who had merely technical or general support participation may be mentioned in the Acknowledgement section.
The kind of contribution from each author to the study performance and to the manuscript preparation must be explained in the following areas, at the moment of submission:
- Study design;
- Data collection, analysis and interpretation;
- Manuscript writing.
Acronyms and Terminology
The use of acronyms must be low. When long expressions need to be repeated, it is recommended that its capital initial letters substitute them after the first mention, which must be followed by the initials between parentheses.
All acronyms in tables and pictures must be defined in their respective legends.
The use of abbreviations must be avoided in Abstracts (in both languages).
Only the generic name of the used medication must be mentioned in the work; the use of commercial names is inadvisable.
SALUS adopts the Universal Official Anatomical Terminology, approved by the International Federation of Associations of Anatomists (IFAF)
PREPARATION OF THE MANUSCRIPT
Sections of the Manuscript
Title and Authors. The work title, in Portuguese and English, must be concise and informative. Each author’s full name, titles and institutional links must be provided.
Abstract (in Portuguese and English). The abstract must be structured in four sections: Objective, Methods, Results and Conclusion. The Abstract in English must follow the same structure of the Abstract in Portuguese. Acronyms must be avoided. The maximum number of words must follow the recommendations of the table (Limits for each article type). The abstract must not be structured in Case Report articles (informative or free). The clinical-surgical correlations and multimedia sections dispense abstract.
Descriptors. Three to five descriptors (keywords) must also be included. The descriptors may be consulted in the electronic address http://decs.bvs.br/, which contains words in Portuguese and English or www.nlm.nih.gov/mesh, for only English words, or in the respective links available at the submission system of the magazine.
Manuscript Body. The Original Articles and Experimental Works must be divided in the following sections: Introduction, Method, Results, Discussion, Conclusion and Acknowledgements (optional). The Case Reports must be structured with the sections: Introduction, Case Report and Discussion; and the Clinical-surgical Correlations must be divided in Clinical Data, Electrocardiogram, Radiogram, Echocardiogram, Diagnosis and Operation. The Multimedia section must present the following sections: Patient Featuring and Description of the Employed Technique. The Review Articles and Special Articles may be structured in sections according to the author’s order.
The Letters to the Editor must, at first, comment, discuss or criticize articles published in SALUS, but they may also refer to other themes of general interest. It is recommended a 1.000 words maximum and it includes five references, maximum, with or without title. Whenever possible, an answer from the authors of articles in discussion will be published along with the letter.
References
The references of printed and electronic documents must be standardized according to the Vancouver style, elaborated by the International Committee of Medical Journal Editors (ICMJE, available at: http://www.icmje.org).
The references must be identified with Arabic numerals in the text, superscript, obeying the citation order in the text. The accuracy of the references is an author’s responsibility. If more than two references are cited in sequence, only the first and the last must be typed, separated by a dash (Example: 6-9). In cases of alternate citation, all the references must be typed separated by comma (Example: 6,7,9).
All authors must be cited in publications with up to 6 authors; in publications with more than 6 authors, the first 6 are cited followed by the Latin expression “et al.”.
Titles of journals must be abbreviated according to the List of Journals Indexed for MEDLINE (available at: http://www.nlm.gov/tsd/serials/lji.html).
References Models
Magazine Article
Issa M, Avezum A, Dantas DC, Almeida AFS, Souza LCB, Sousa AGMR. Fatores de risco pré, intra e pós-operatórios para mortalidade hospitalar em pacientes submetidos à cirurgia de aorta. Rev Bras Cir Cardiovasc. 2013; 28(1):10-21.
Organization as Author
Diabetes Prevention Program Research Group. Hypertension, insulin, and proinsulin in participants with impaired glucose tolerance. Hypertension. 2002;40(5):679-86.
Without indication of authorship
21st century heart solution may have a sting in the tail. BMJ. 2002;325(7357):184.
Article published electronically before the print version (“ahead of print”)
Atluri P, Goldstone AB, Fairman AS, Macarthur JW, Shudo Y, Cohen JE, et al. Predicting right ventricular failure in the modern, continuous flow left ventricular assist device era. Ann Thorac Surg. 2013 Jun 21. [Epub ahead of print]
Article of Internet Journal
Machado MN, Nakazone MA, Murad-Junior JA, Maia LN. Surgical treatment for infective endocarditis and hospital mortality in a Brazilian single-center. Rev Bras Cir Cardiovasc [online]. 2013[cited 2013 Jun 25];28(1):29-35. Available at: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0102-76382013000100006&lng=en&nrm=iso
Book Chapter
Chai PJ. Intraoperative myocardial protection. In: Mavroudis C, Backer C, eds. Pediatric cardiac surgery. 4th ed. Chichester: Wiley-Blackwell; 2013. p.214-24.
Book
Cohn LH. Cardiac surgery in the adult. 4th ed. New York: McGraw-Hill;2012. p.1472.
Thesis
Dalva M. Estudo do remodelamento ventricular e dos anéis valvares na cardiomiopatia dilatada: avaliação anátomo-patológica [Tese de doutorado]. São Paulo: Universidade de São Paulo, 2011. 101p.
Legislation
Conselho Nacional de Saúde. Resolução n. 466, de 12 de dezembro de 2012. Dispõe sobre diretrizes e normas regulamentadoras de pesquisas envolvendo seres humanos. Bioética. 1996;4(2 Supl):15-25.
Conselho Nacional de Controle de Experimentação Animal. Resoluções n. 12 e 13, de 20 de setembro de 2013. Dispõem sobre a diretrizes brasileiras para o cuidado e a utilização de animais para fins científicos e didáticos (DBCA) e prática de eutanásia.
Other examples of references may be consulted at the website:
http://www.nlm.nih.gov/bsd/uniform_requirements.html
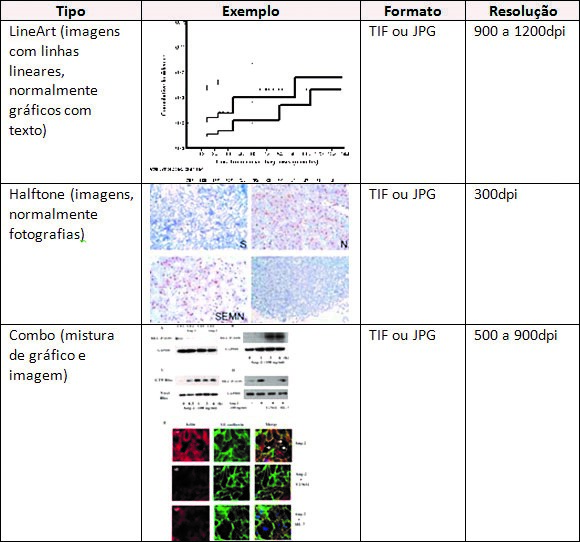
Tables and Pictures
The Tables and Pictures must be numbered according to order appearance in the text, contain a title and be in separate files. The tables must not contain redundant data, previously mentioned in the text. They must be open in the sides and with a totally white background.
The acronyms used in the tables must be mentioned in alphabetical order, in the footnote, with their respective full forms. Similarly, the acronym used in pictures must be explained in the legends.
The pictures will only be published in colors if the author agrees to afford the costs of impression of colorful pages.
Only images in formats TIFF or JPEG will be accepted, with minimum resolution according to the kind of image, for both black and white and colorful images, according to the Table below:
SALUS requests that authors save the original images with them, because if the images submitted online present any hindrance for printing, we will get in touch so they can send us the originals.
Limits per Type of Article
The following criteria sorted by type of publication must be observed, aiming to rationalize the space in the magazine and allow more articles per edition. The electronic word count must include the initial page, abstract, text, references and picture legends. The titles have 100 characters limit (counting the spaces) for Original Articles, Review and Updating Articles and Experimental Work; the other categories have 80 characters (counting the spaces) title limit.
| Original
Article |
Editorial | Review / Updating Article | Case Report | Case Report and Literature Review | Brief Communication and Preliminary Note | Letter to the Editor | Experimental Work | Clinical-Surgical Correlation | Multimedia | |
| Maximum number of authors |
10 |
4 | 5 | 4 |
6 |
4 | 2 | 10 | 4 | 4 |
| Abstract – Maximum number of words | 250 | - | 200 | 100 | 100 | 100 | - | 250 | - | - |
| Maximum number of words | 6,000 | 1,000 | 8,000 | 1,500 |
3,000 |
2,000 | 400 | 6,000 | 800 | 800 |
| Maximum number of references | 40 | 10 | 75 | 6 |
20 |
6 | 6 | 40 | 10 | 10 |
| Number of tables and pictures | 8 | 2 | 8 | 2 |
6 |
2 | 1 | 8 | 2 | 1 |
| Abbreviated Title | — | — | — | — | — | 40 Characters | — | — | — | — |
Table Model:
Picture Model:
Check it before sending the work
- Submission letter indicating the manuscript category.
- Statement by authors and co-authors saying that they agree with the content of the manuscript.
- Approval Letter by the Ethics Committee of Research.
- Manuscript written in software Word 97 or above (formatted to A4); font size 12, space 1.5, font Times New Roman; paged; mathematical symbols and Greek characters using font Symbol.
- Manuscript in the limits adopted by SALUS for its category.